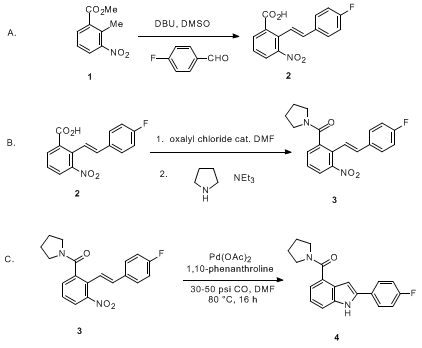
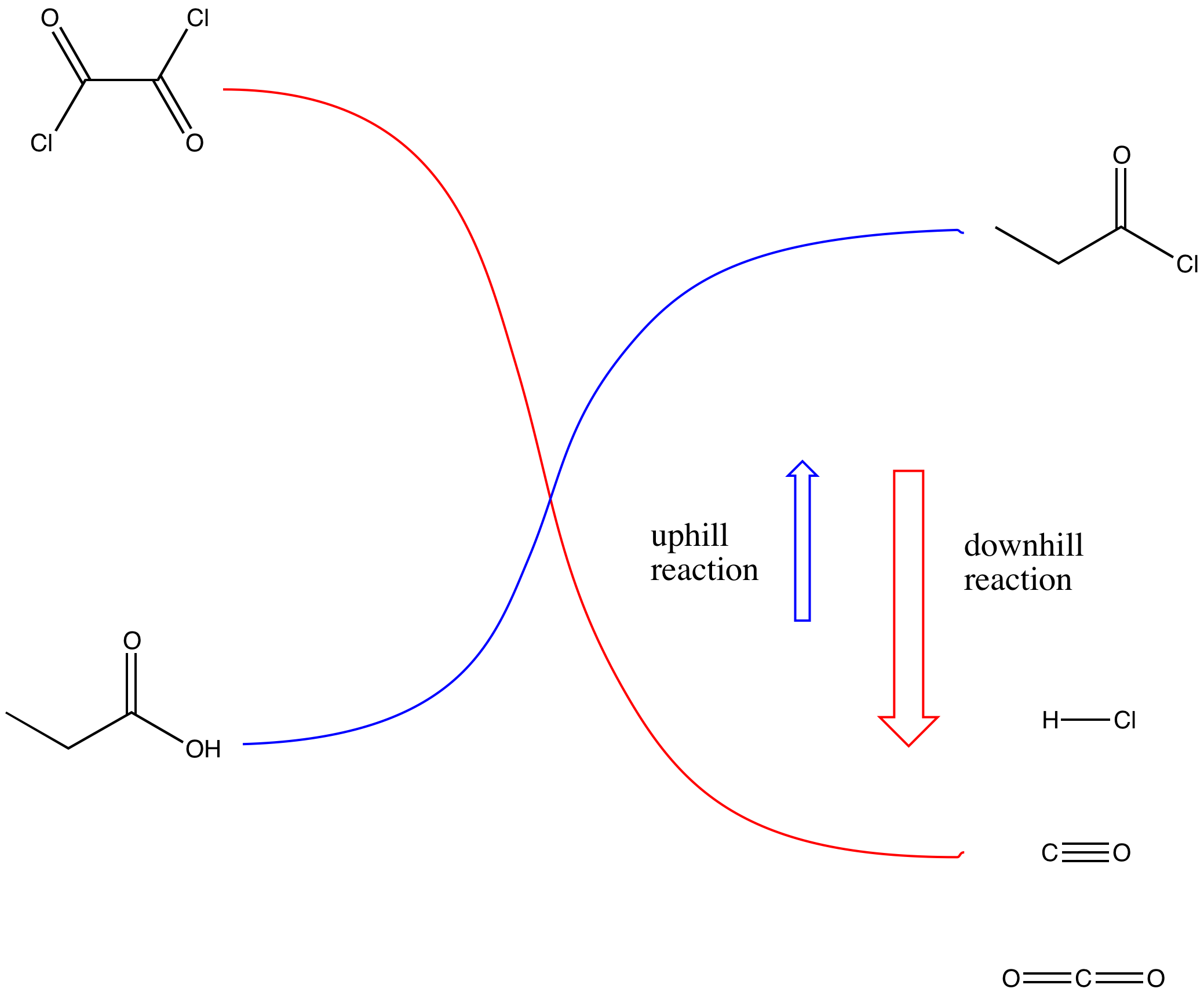
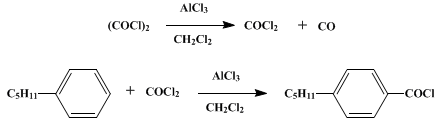
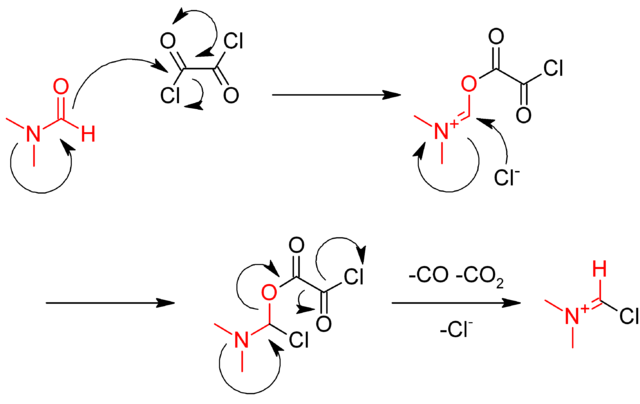
Alcohols as Latent Coupling Fragments for Metallaphotoredox Catalysis: sp3–sp2 Cross-Coupling of Oxalates with Aryl Halides | Journal of the American Chemical Society

Oxalyl Chloride: A Versatile Reagent in Organic Transformations - Mohammadkhani - 2019 - ChemistrySelect - Wiley Online Library

Stereoretentive cross-coupling of chiral amino acid chlorides and hydrocarbons through mechanistically controlled Ni/Ir photoredox catalysis | Nature Communications

Molecules | Free Full-Text | Synthesis of Anti-Inflammatory Spirostene-Pyrazole Conjugates by a Consecutive Multicomponent Reaction of Diosgenin with Oxalyl Chloride, Arylalkynes and Hydrazines or Hydrazones

organic chemistry - Synthesis of oxalyl chloride that doesn't involve phosphorus - Chemistry Stack Exchange

Scheme 1. Synthesis of (E)-I-(E)-III. (a) Oxalyl chloride, CH 2 Cl 2 ;... | Download Scientific Diagram

Oxalyl Chloride: A Versatile Reagent in Organic Transformations - Mohammadkhani - 2019 - ChemistrySelect - Wiley Online Library

Solvent-free synthesis of substituted five membered heterocycles: One-pot reaction of primary amine and alkyl propiolate or isothiocyanate in the presence of oxalyl chloride - ScienceDirect